this post was submitted on 02 Dec 2024
642 points (95.6% liked)
Science Memes
11287 readers
2610 users here now
Welcome to c/science_memes @ Mander.xyz!
A place for majestic STEMLORD peacocking, as well as memes about the realities of working in a lab.

Rules
- Don't throw mud. Behave like an intellectual and remember the human.
- Keep it rooted (on topic).
- No spam.
- Infographics welcome, get schooled.
This is a science community. We use the Dawkins definition of meme.
Research Committee
Other Mander Communities
Science and Research
Biology and Life Sciences
- [email protected]
- [email protected]
- [email protected]
- [email protected]
- [email protected]
- [email protected]
- [email protected]
- [email protected]
- [email protected]
- [email protected]
- [email protected]
- [email protected]
- [email protected]
- [email protected]
- [email protected]
- [email protected]
- [email protected]
- [email protected]
- [email protected]
- [email protected]
- [email protected]
- [email protected]
- [email protected]
- [email protected]
- !reptiles and [email protected]
Physical Sciences
- [email protected]
- [email protected]
- [email protected]
- [email protected]
- [email protected]
- [email protected]
- [email protected]
- [email protected]
- [email protected]
Humanities and Social Sciences
Practical and Applied Sciences
- !exercise-and [email protected]
- [email protected]
- !self [email protected]
- [email protected]
- [email protected]
- [email protected]
Memes
Miscellaneous
founded 2 years ago
MODERATORS
you are viewing a single comment's thread
view the rest of the comments
view the rest of the comments
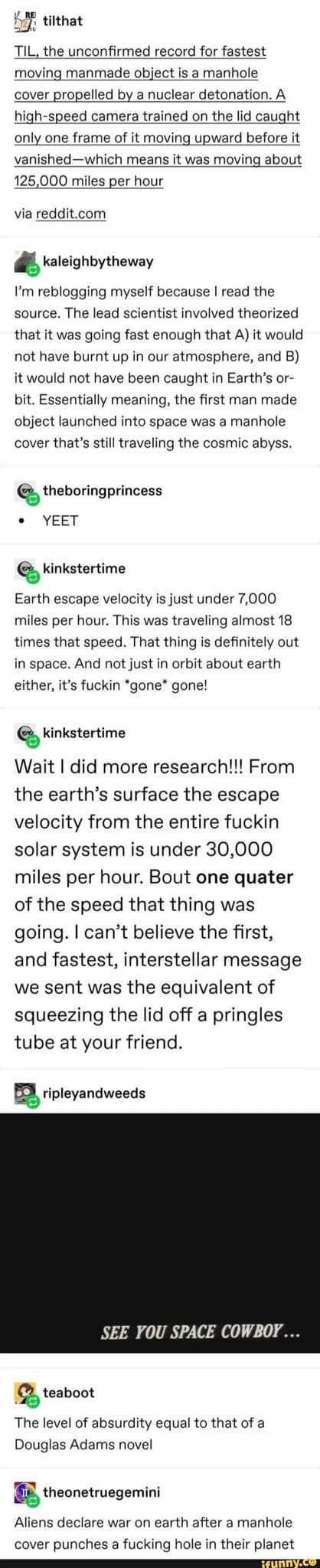
Sadly, the cover likely did burn up in the atmosphere at those speeds, like a meteorite in reverse.
I'm not so sure.
Let's compare with the Apollo Command Module heat shield, a remarkably close analogue for the bore cap. They're a similar weight (3,000 lb for the heat shield, 2,000 lb for the bore cap) and have melting points within an order of magnitude of each other (5,000°F for the AVCOAT heat shield and about 2,800°F for the iron bore cap). They're even both of a similar shape and aerodynamic profile (disc-shaped and blunt). Both had to travel 62 miles (the distance from sea level to the Karman Line, where atmosphere becomes negligible).
The Apollo CM made that distance in about seven minutes; at 130,000mph, the Pascal B bore cap took at most 1.72 seconds to make the trip.
What was discovered during the development of the Apollo heat shield is that the blunt shape caused a layer of air to build up in front of the spacecraft, which reduced the amount of heating that convected into the heat shield directly. This reduced the amount of heat load that the heat shield needed to bear up under.
Further, it's also worth noting that the Apollo command modules weren't tumbling, which the bore cap likely would have been, allowing brief instants during its ascent for the metal to cool before being subjected again to the heat of the ascent.
But probably most critical at all is the remarkably brief amount of time that the bore cap spent in atmosphere. This person did the math on how much power it would take to vaporize a cubic meter of iron, and the answer is 25,895,319 kJ. Now, the bore cap isn't quite a cubic meter, but we can use all of his calculations and just swap in 907kg (2000lbs):
To heat the bore cap to iron's melting point: 0.46 kJ/kg * 907 kg * (1808K-298K) = 630,002 kJ
To phase change the iron from solid to liquid: 69.1 KJ/kg * 907 kg = 62,674 kJ
To heat the bore cap to iron's boiling point: 0.82 kJ/kg * 907 kg * (3023K-1808K) = 903,644 kJ
To phase change the iron from liquid to gas: 1520 kJ/kg * 907 kg = 1,378,649 kJ
So, in total, 2,974,969 kJ. The Apollo heat shield encountered a peak of 11,000 kJ/m^2/s. Since the Pascal B bore cap was about a meter in diameter and was traveling through the atmosphere for about two seconds, we can very neatly estimate that it absorbed a maximum of 22,000 kJ due to atmospheric compression--not even close to enough to get it to melting temperature.
Interestingly, early missiles actually did use solid metal heat shields; not iron, but titanium, beryllium, and copper. They were effective, but abandoned due to their weight.
I don't think you can compare the Apollo heat shields to a bore cap being launched into space. For one thing, the Apollo shield started in the very thin upper atmosphere, and they came in at an angle that meant they bled off as much speed/energy as possible in that thin upper atmosphere before going into the thicker atmosphere. In fact, one of the engineers said that if they came in too steep they'd generate too much heat and probably not survive the re-entry.
The layer of air you're talking about at the front of the spacecraft was what heated up the heat shield. Instead of causing heating via friction, the heat was the result of compressing the air. The amount of compression you're talking about would be orders of magnitude higher for something starting at 40 km/s in the thick lower atmosphere.
Also, the Apollo heat shield did heat up to 5000F or 2800C but was designed to be ablative, so that the hot layers burned off and flew off to the sides leaving new material to be heated up and burned off. This concrete and metal plug wouldn't have been designed the same way. Concrete apparently melts at 1200C, and steel is approximately the same, so it's very likely some of it melted or vaporized, the question is how much.
I don't know where you're getting the maximum of 22MJ of energy. The whole point of Apollo not going directly into the atmosphere was to take as long as possible to slow down, going through the thinnest part of the atmosphere for as long as possible. The whole point would be to reduce their energy-per-second as low as possible by taking as many seconds as possible. One reasonable first approximation of the energy would be to integrate the entire energy per second / power for Apollo's re-entry over the entire 7 minutes (or however long it took until parachutes deployed) and then divide that energy by 2 for the 2 seconds the plug was in the atmosphere.
My guess is that that would have been temperatures well in excess of 1200C which would have made the outer surface start to melt, and most likely a temperature where it just turns to plasma. Would it all have melted / vaporized / plasmafied away? I don't know, it's a huge plug. Since it was launched vertically, anything remaining would probably have come right back down. But, that's assuming it stayed in one piece. I'm guessing it broke apart due to the stresses on it, and breaking apart would have meant more surface area, which would have meant more areas exposed to massive heating, which would have meant more breaking apart.
TL;DR: I doubt it made it out of the atmosphere.
I don't know that that makes a huge difference to the physics involved, though it certainly may have.
But in that case we're talking about human survivability, and a chunk of solid iron is going to survive a whole lot longer than humans or delicate instrumentation. It might look a little worse for the wear, but it's much more likely to be recognizable after the whole experience than anything designed for people.
But after initial heating, the air cushion begins heating itself up instead of the object, reducing the amount of heat the object receives.
But it would also tail off as the bore cap heated, reducing stresses on it as it went higher.
True, but on the other hand the a Apollo heat shield wasn't designed to convect heat to other parts of itself. And again, it had a much harder job (keep the Apollo command module at human-survivable temperatures) than the bore cap (not reach the boiling point of iron).
All the stuff I read only mentioned the iron, but keep in mind that it has to not only reach the melting point but also undergo phase change, which requires a lot more energy.
11 kJ per m² per second was the peak amount of energy that the Apollo heat shield encountered. Double that for the approximately two seconds it would've been in atmosphere, and it's a pretty handy approximation since the bore cap was about a meter itself.
You're right, the total amount would've been a way better approximation than the peak. Worth looking into.
I don't have any argument with that. I think the outer surface would definitely have begun to melt.
Yep. Even just considering the amount of time it would take for the heat to excite all the molecules in the massive chunk of iron, and then for them all to undergo phase change, I just don't think it could've made it.
That's something I couldn't find information on: is iron's tensile strength high enough to prevent the thing shattering apart on contact with air? I'm inclined to think it is—chunks of meteorites bigger than a meter have made it through the atmosphere, for instance. The Hoba meteorite is estimated to only be slightly bigger than it is now before its atmospheric entry, and it's way bigger than the bore cap. Similar composition, too.
Either way, I like researching it.
Edit: also, the bore cap starting at the bottom of the atmosphere means that it's likely it experienced less fracture stress, since the air would've accelerated with it rather than being static.
Of course it will make a difference. The whole challenge is about managing the heat build-up, which is the energy per second (i.e. power). If you hit the thin upper atmosphere you're encountering less material, so less friction / pressure, so less heating. It means you can keep the heat on the heat shield in a manageable range, rather than putting it at a temperature where it would melt or explode.
No, both heat up. The air cushion transfers its heat to the object next to it. At the kinds of pressures we're talking about, you might even be getting nitrogen plasma rather than just nitrogen gas.
If it went high enough for that to matter. If it disintegrated in the lower atmosphere it wouldn't matter that the air got thinner in the upper atmosphere.
Is a metre the original size, or the final size? Also, reverse meteors (something starting with its maximum speed in the lower atmosphere) are doing things the hard way. Rather than getting slowed down initially by the thin upper atmosphere and then only hitting the thick atmosphere once they're slower, they start out in the thickest atmosphere. OTOH, a meteor is a random collection of rock and metal formed by gravity in space. A pure metal plug cast on Earth is probably going to be a lot less prone to breaking apart.
That doesn't make sense to me. Something in a thicker medium is going to experience more stress. Try pushing a cracker through the air vs. through water vs. through gelatin. Which medium will cause the cracker to crack first? Obviously it's the thicker medium.
Most of this is going to be "eh, agree to disagree" because we just don't have enough data. But I do want to call out a couple of things:
Over time, yes. But the bore cap doesn't have very much of it. Heat transfer is not instantaneous; would it be long enough for the air to transfer its heat to the object, before the object reaches the Karman Line? Radiation is pretty quick (like, speed-of-light quick), but conduction is much slower; particularly when one of the bodies (the air) is an insulator. And with iron being an excellent conductor, any heat transferred will be spread throughout the body more quickly than it can be absorbed.
True, but it's not like there's a line (er, well, I mean, not a physical demarcation...there is the Karman Line, but...ah, you know what I mean). Atmospheric density is a decreasing gradient from the ground to the Karman Line. So as it approaches its mechanical and physical limits, the amount of energy acting upon it decreases millisecond by millisecond. Is that enough to save it? Shrug. Not enough data. But it's possible.
Actually it's almost three meters, and as far as we can guess that was about its original size. Though in fairness, it was entering the atmosphere at a steeper angle and may even have come down entirely in "dark flight." Still, there are other large meteorites which have impacted at a size greater than 1 meter across, though obviously we have no way to confirm exactly how big they were before they landed.
True! But remember, the "reverse meteor" (great phrase, btw) is not hitting the stationary atmosphere at full speed like a regular meteor (or space capsule) does. The iron plug accelerated (incredibly quickly, but it did accelerate) while already in contact with the air above it. This means that the air accelerated at the same rate the iron did, reducing the fracture forces that would seek to crack it. Imagine the difference between swishing your hand in a swimming pool vs. slapping the surface of a swimming pool; it may require more force, but it won't hurt as badly.
Oh, great point, and one I hadn't thought about. Something that's an aggregate of 80% iron and 20% "other stuff" isn't going to have nearly as much tensile strength as a homogeneous plate of iron.
I'm not so sure... At those speeds, it would've taken under 10 seconds to completely clear the atmosphere. Even with intense compressional heating, I don't think it would've been in contact with the atmosphere long enough to completely vaporize — although it probably didn't look much like a manhole cover anymore by the time it escaped.
I don't think melting is the issue here. I think it literally disintegrates at those speeds. Like, this is Mass Effect mass driver level of impact with the atmosphere.
For reference, RICK ROBINSON'S FIRST LAW OF SPACE COMBAT: "An object impacting at 3 km/sec delivers kinetic energy equal to its mass in TNT."
Assuming the lid is travelling 55km/s, it's well beyond that point. The atmosphere it's travelling through is basically a solid at that speed. Even if it isn't heating due to the friction (and waiting for heat flow), it is heating due to the compressive force of being slammed into the atmosphere. It's very likely the whole thing vaporized.
But I could be wrong, and some alien SOB is going to have a bad day when the manhole cover slams into their ship in interstellar space.
Would vaporization slow the material though? Perhaps the end result wasn't a manhole escaping the solar system but a huge collection of microscopic metal fragments scattershot that direction. Which really makes the Mass Effect quote even more relevant to a huge amount of aliens somewhere.
Vaporization would certainly slow the material. It's transitioning kinetic energy into thermal.
Also, the vaporized iron would disperse outward rather than stay coherent.
It would spread outward a bit, but the entire kinetic energy and momentum in the system would remain the same. But, the more it broke apart, the more surface area it would have. The more surface area, the more surface exposed to heating. The more heating, the more it would break apart. I'm guessing that it was a silicon, iron and oxygen plasma without individual grains by the time it hit the upper atmosphere.
The atmosphere is just about 10 kg/m^2 in sectional density; the manhole cover was very likely higher than that, wouldn't that mean the cover's mass should have come out at the other side, intact or not?
It was being propelled by a nuclear blast. The speed was calculated from 1 frame of a high speed camera. It most definitely vaporized.
And for reference, the earth escape velocity from the surface is 11.2 km/s or 25,000 mph, not 7,000 mph.
To escape the solar system from the earth surface, the minimum speed is 16.6 km/s, or 37,100 mph. But this assumes that you launch in the correct direction to take the most advantage of the Earth's 30 km/s. If you launch in the most disadvantageous direction, you can add another 60 km/s to escape.
So sad about what happened to planet Kerbin.
Yes, it absolutely would have vaporized before exiting the atmosphere.
Here's a video on the subject: https://www.youtube.com/watch?v=mntddpL8eKE